By A Mystery Man Writer

Standard Operating Procedure (SOP) and Guideline for the Receipt, Storage, Preparation, Growth Promotion Test, use, and Disposal of microbiological media.

How To Establish Growth Promotion Tests For Pharmaceutical Culture Media
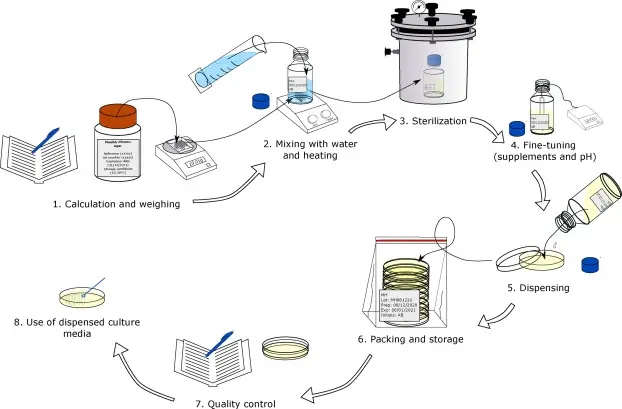
Best Practices for Preparation of Culture Media • Microbe Online

The Ultimate Guide to Good Laboratory Practices (GLP)

What is GMP, Good Manufacturing Practices
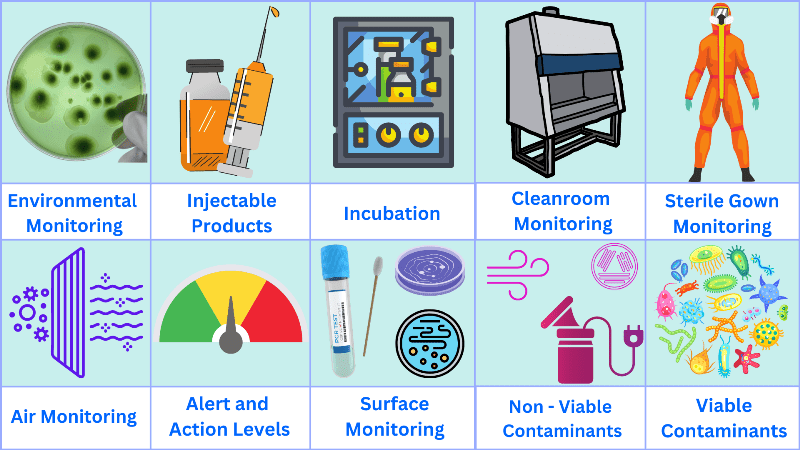
What is environmental monitoring in pharmaceutical industry

EU GMP Annex 1
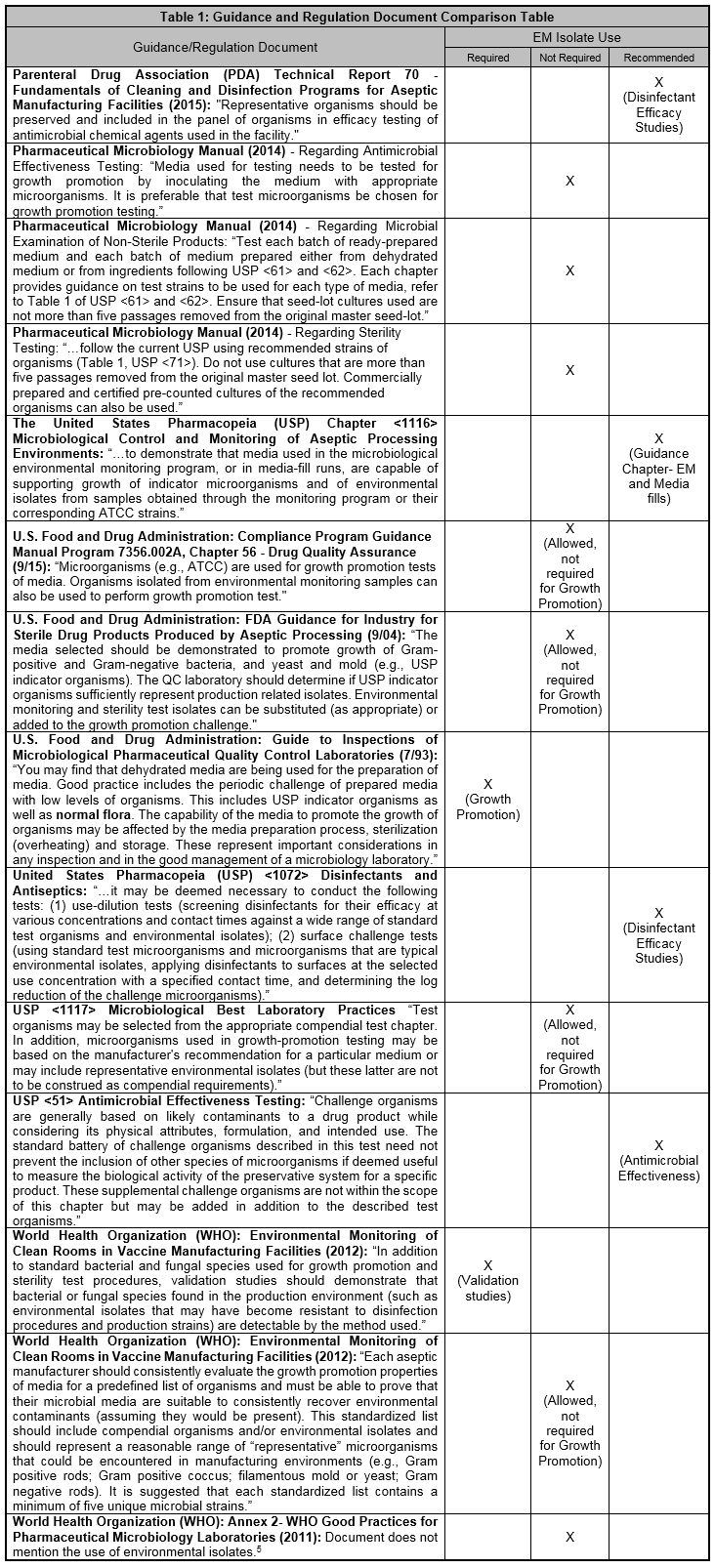
Environmental Isolates What's The Proper Use Of In-House Cultures
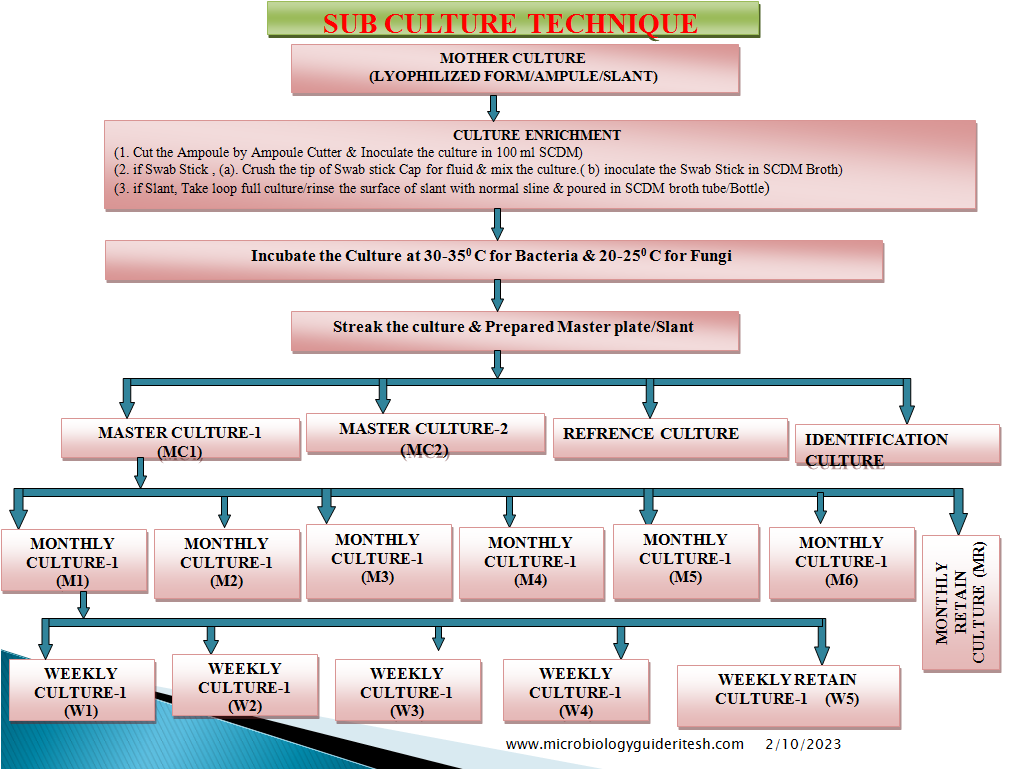
SOP - Microbiology Guidelines

Entry and Exit of Micro Lab Sop, PDF, Clothing

PDF) Microbiological Culture Media: A Complete Guide for Pharmaceutical and Healthcare Manufacturers

Organoids, Free Full-Text
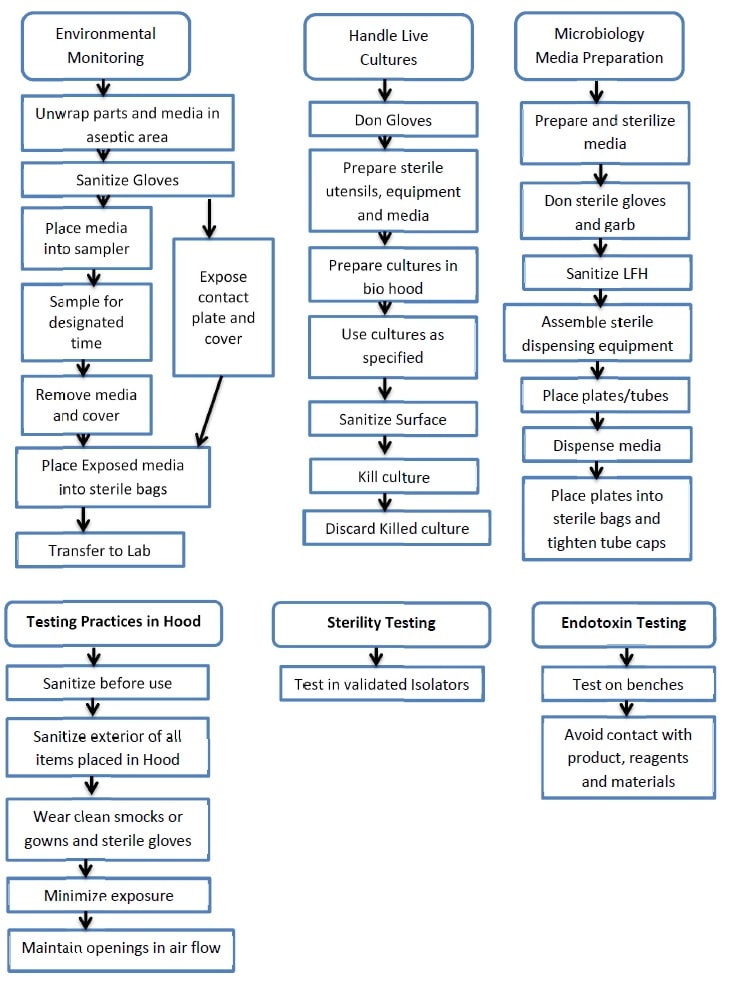
Aseptic Technique for Microbiological Testing - Pharma Beginners
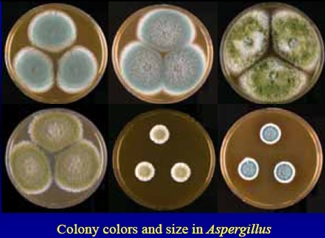
SOP for Isolation and Identification of Microorganisms - Pharma Beginners

PDF) Microbiological Culture Media: A Complete Guide for Pharmaceutical and Healthcare Manufacturers

SOP for Isolation and Identification of Microorganisms - Pharma Beginners