SOLVED: For a gas at a given temperature, the compression factor is described by the empirical equation: z = 1 - 8.50 × 10^(-3)P/P° + 3.50 × 10^(-5)(P/P°)^2 where P° = 1
By A Mystery Man Writer

VIDEO ANSWER: Hello students: let's look at the question: l n, that integrate integration and 0 z minus 1 bracket, close d p by p here. Minus 1 is equal to minus 8.50 into 10 to the power minus 3 p by p, not plus 3.50 into 10. To the power minus 9. P
Numerade is a venture-backed, high-growth education technology startup based in Pasadena. We are singularly focused on creating exceptional video and interactive content experiences for education making the knowledge and skills of world class educators widely accessible and affordable to student audiences of all backgrounds. Our mission is to close the educational opportunity gap by unlocking and democratizing access to extraordinary educators and the content they have to offer.

OneClass: For a gas at a given temperature, the compression factor is described by the empirical equa

COMPRESSIBILITY factor Z, Using P and v in 3 Minutes!

Introduction to Fluid Mechanics and Fluid Machines [2 ed.] 0070667624, 9780070667624

SOLVED: For a gas at a given temperature, the compression factor is described by the empirical equation: z = 1 - 8.50 × 10^(-3)P/P° + 3.50 × 10^(-5)(P/P°)^2 where P° = 1

Walpole 8 probabilidad y estadística para ciencias e ingenierias parte2 by Marco Acuña - Issuu

Solved For a gas at a given temperature, the compression

Physical Chemistry The Compression Factor (Z) [w/1 example]
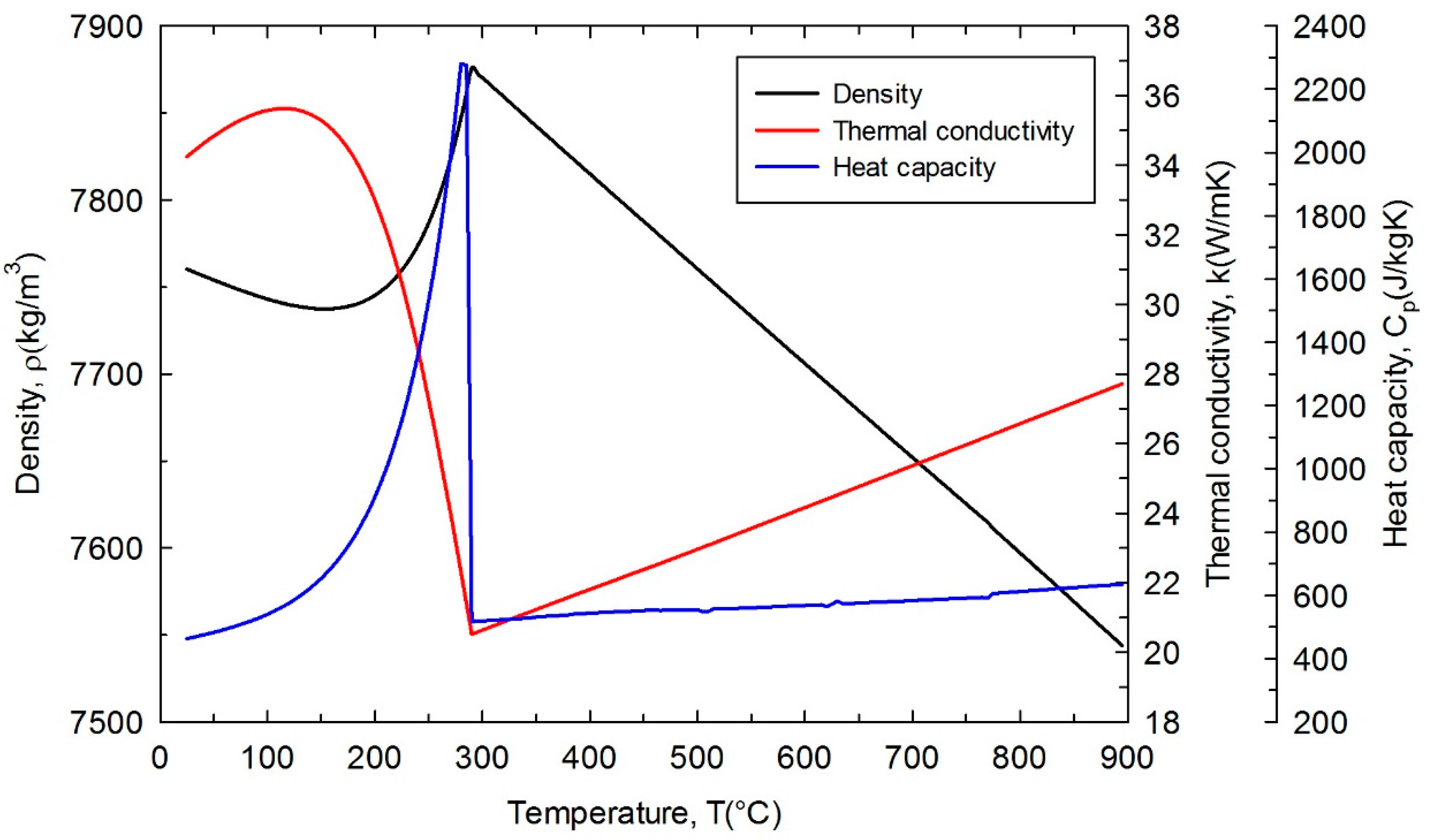
Metals, Free Full-Text

Thermodynamics - 3-7 Ideal Gas Equation with compressibility factor

As a first approximation, the compression factor, Z
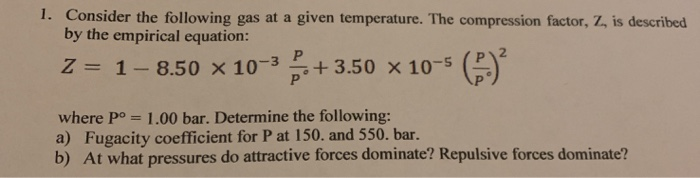
Solved 1. Consider the following gas at a given temperature.

Solved - Evaluate the compressibility factor of two

SOLVED: Use our numerical knowledge to solve all of the following questions: Problem (6 points) The Van der Waals equation of state for vapor is: (P + a/v^2)(v - b) = RT

Computational Physics