An ideal gas is taken from (Pi , Vi ) to (Pi , Vi ) in three different ways. Identify the process in (d) Equal work is done in Process A, B & C

An ideal gas initially P_i ,V_i , and T_i is taken through a cycle as shown in Figure. (a) Find the net work done on the gas per cycle 1.00 mol of

An ideal gas is taken from state A to the state B, as shown in the P V diagram. The work done in the process is

SOLVED: 3P; Pi A V 3V; A 1 mole of ideal gas initially at Pi-l Pa, Vi–5 m, and Ti= 0°C is taken through a cycle as shown in the above Figure.
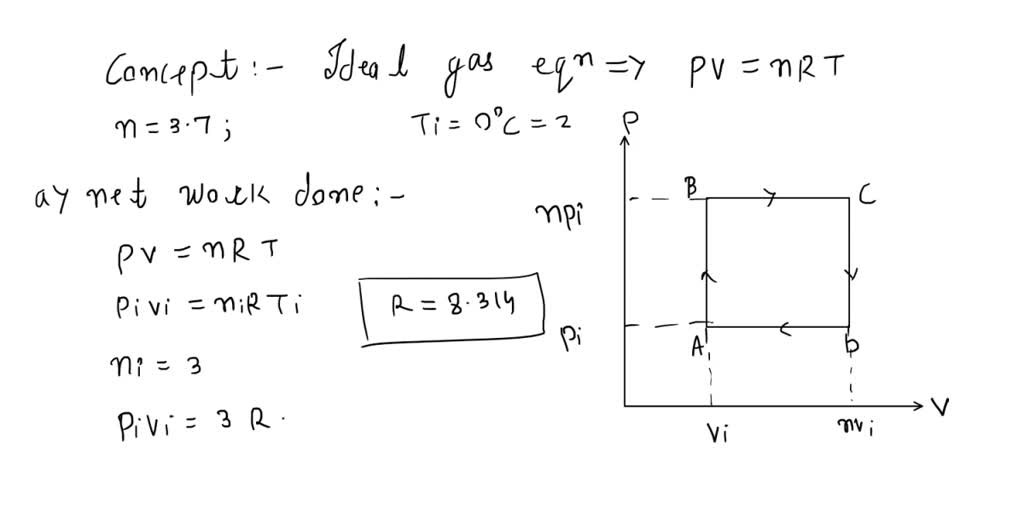
SOLVED: ideal gas initially at Pi, Vi, and Ti is taken through cycle as shown below: (Let the factor n 3.7.) nf Find the net work done on the gas per cycle
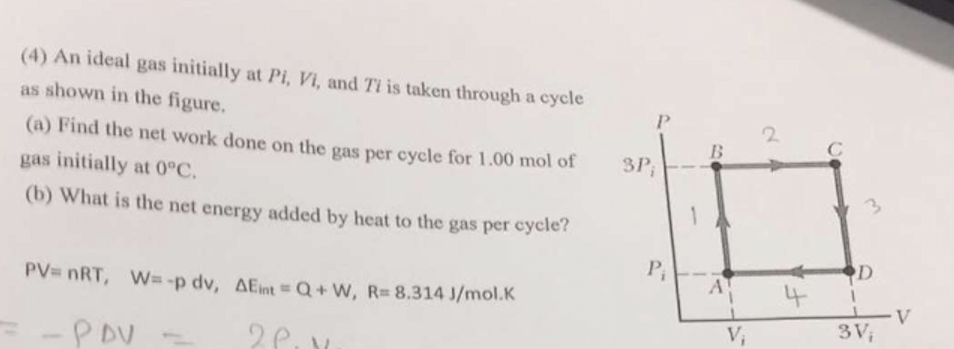
Solved (4) An ideal gas initially at Pi, Vi, and Ti is taken

An ideal gas is taken from state A Pressure P, Volume V to the state B Pressure P/2, Volume 2V along a straight line path in PV diagram as shown in the

Solved An ideal gas initially at Pi, Vi, and Ti is taken
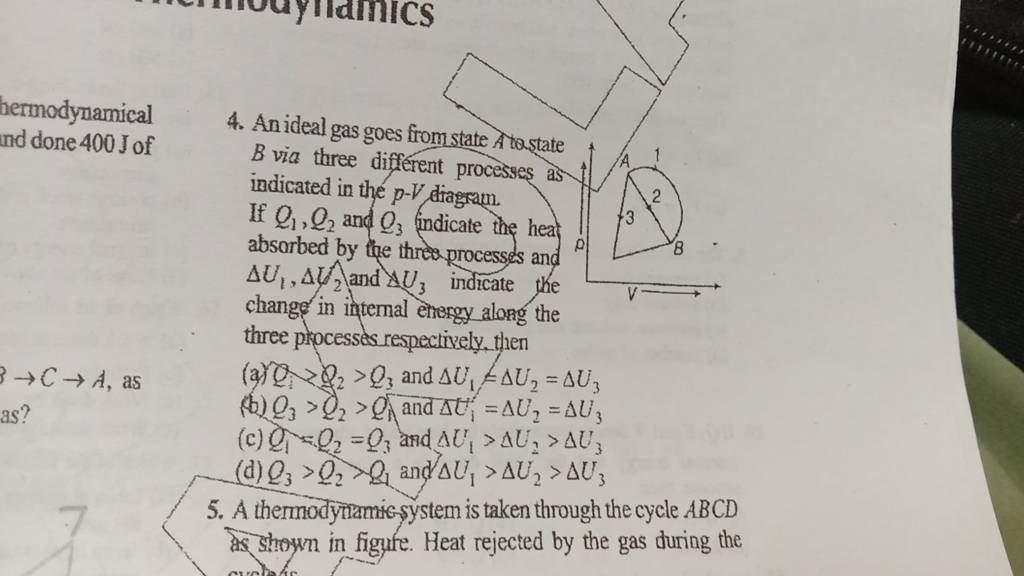
An ideal gas goes from state A to.state B via three different processes a..

An ideal gas is taken from state A to state B via three different processes as shown in the pressure volume (P-V) diagram. If Q4, Q, & Q, indicates the heat absorbed

One mole of a diatomic ideal gas (gamma=1.4) is taken through a cyclic process starting from poi

Solved A system of ideal gas can be taken from state 3 to

One mole of an ideal gas is taken from state A to state B by three different processes a A C B, b A D B and c A E B as