By A Mystery Man Writer
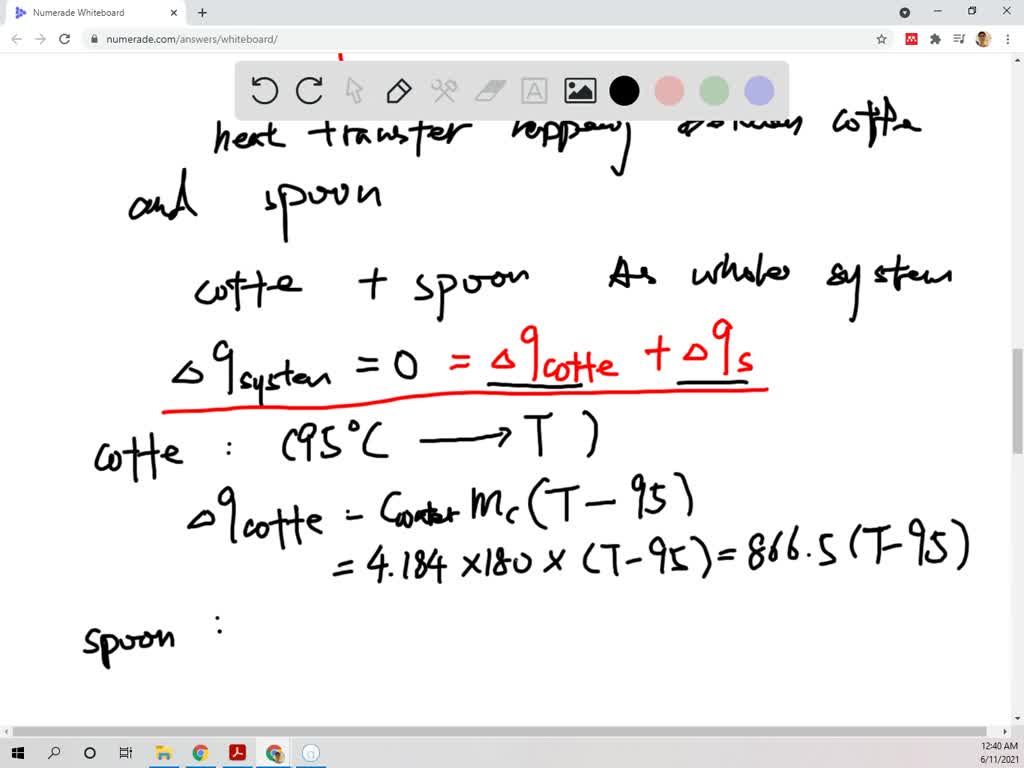
VIDEO ANSWER: Okay. So still another he transferred problem. So in this time ask how much will the temperature of the cop of the coffee up 95°C be reduced When the 45 Grand Silver Spring right is placed in the coff
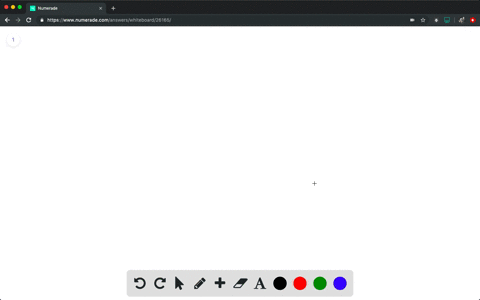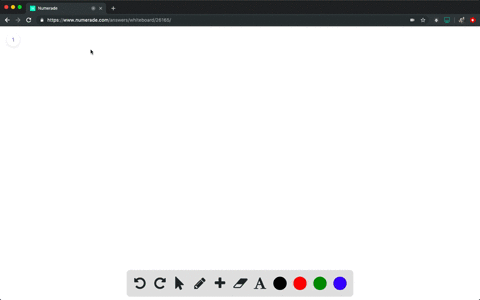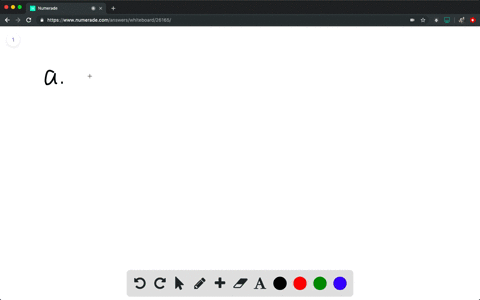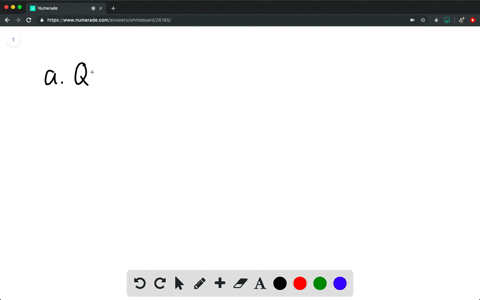
How much will the temperature of a cup (180 g) of coffee at 95 ^ C be reduced when a 45 g silver spoon (specific heat 0.24 J / g^∘ C ) at 25^∘ C is placed in the coffee and the two are allowed to reach the same temperature? Assume that the coffee has the same density and specific heat as water.
Numerade is a venture-backed, high-growth education technology startup based in Pasadena. We are singularly focused on creating exceptional video and interactive content experiences for education making the knowledge and skills of world class educators widely accessible and affordable to student audiences of all backgrounds. Our mission is to close the educational opportunity gap by unlocking and democratizing access to extraordinary educators and the content they have to offer.
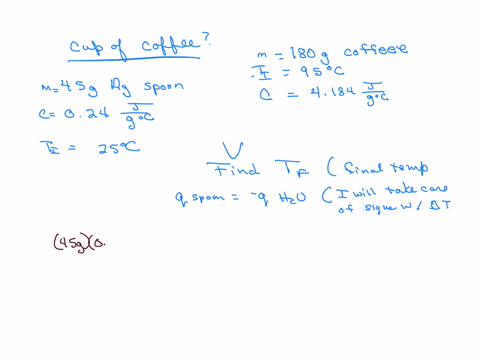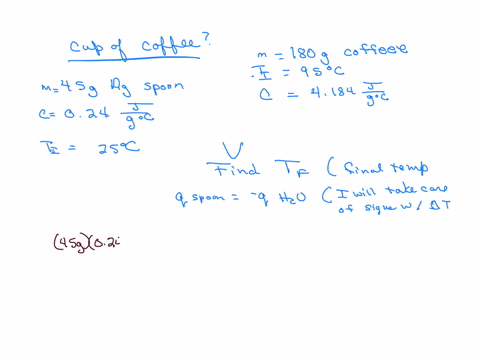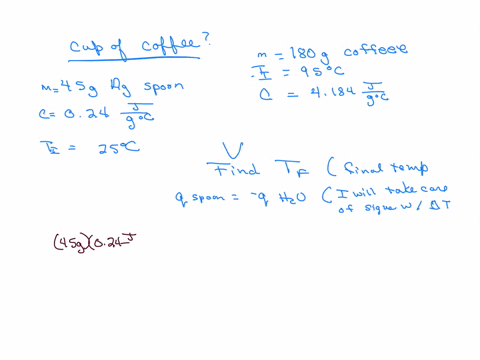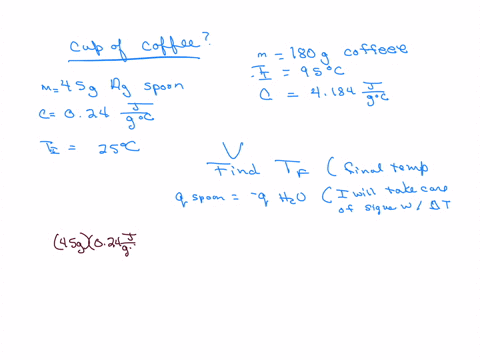
⏩SOLVED:How much will the temperature of a cup (180 g) of coffee
⏩SOLVED:(II) When a 290-g piece of iron at 180^is placed in a 95
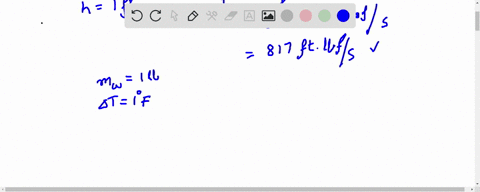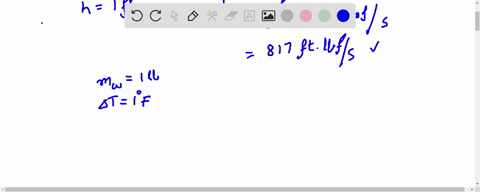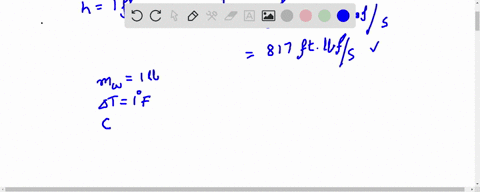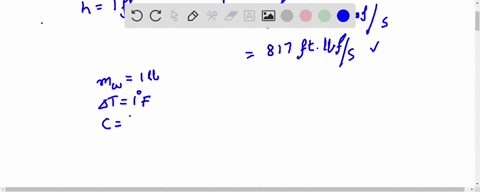
⏩SOLVED:You have 300 g of coffee at 55^∘ C. Coffee has the same
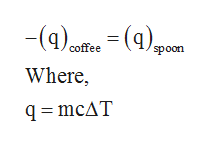
Answered: How much will the temperature of a cup…

⏩SOLVED:In an effort to stay awake for an all-night study session

Solved In the laboratory a coffee cup calorimeter, or
How much will the temperature of a cup (180 g) of coffee at 95 °C
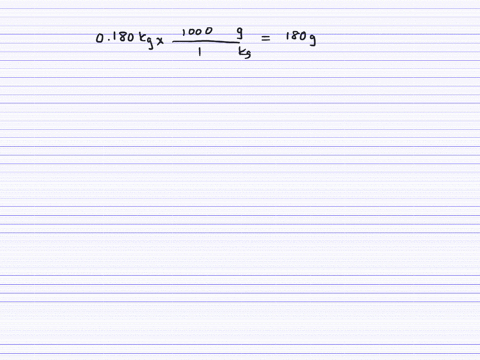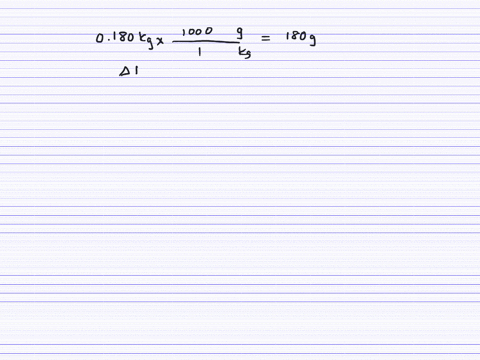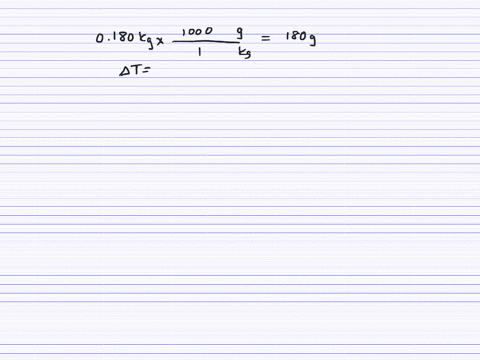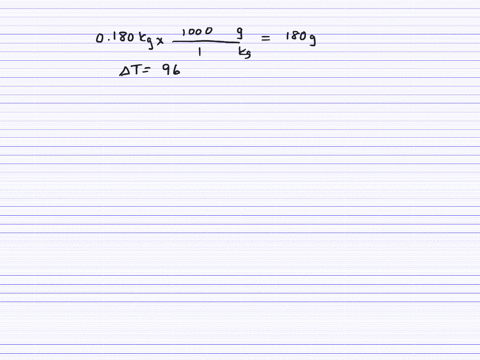
⏩SOLVED:You wish to heat water to make coffee. How much heat (in

⏩SOLVED:A cup of coffee is heated to 180^∘ F and placed in a
Solved A cup contains 193 g of coffee at 93.3 °C. Suppose
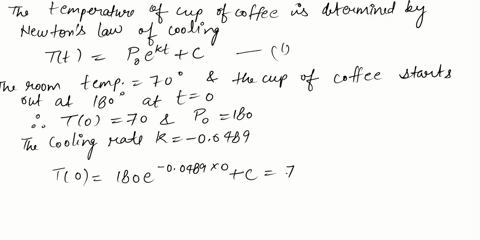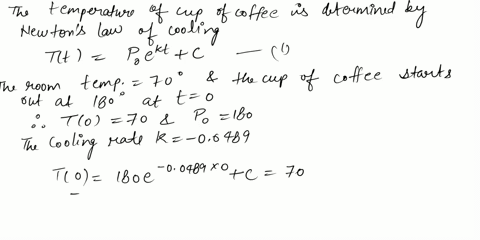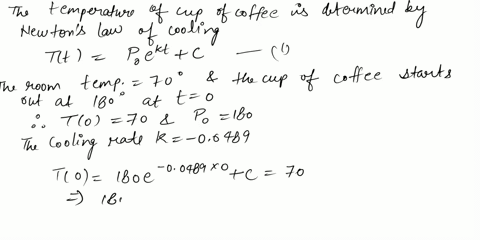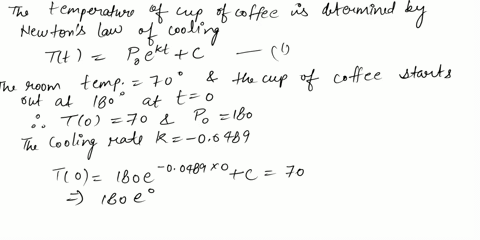
⏩SOLVED:Use Newton's Law of Cooling. A cup of coffee with…

Solved 2. A coffee-cup calorimeter of the type shown in

Solved A coffee cup calorimeter contains 151.41 g of water

⏩SOLVED:How much will the temperature of a cup (180 g) of coffee
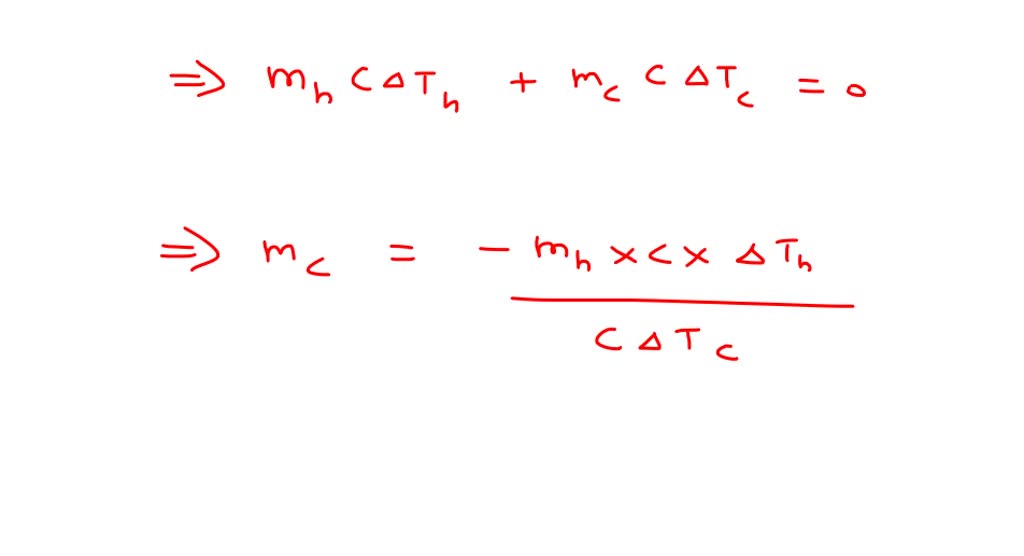
⏩SOLVED:You have 300 g of coffee at 55^∘ C. Coffee has the same